stemintegrado.com
stemintegrado.com
🎯 Clear and challenging objectives.
- General: Build and test a home battery with potatoes or lemons to light an LED or measure voltage on a multimeter.
- Personal: Optimize the design (fruit type, number of cells, electrode size) to achieve the highest possible stable voltage.
🌍 Simple and fun introduction to theory
A battery transforms chemical energy into electrical energy. In this experiment, the acidic lemon pulp (or potato juice) acts as an antioxidant. electrolyte. Two dissimilar metals (e.g., zinc y copper) function as electrodes. Thanks to a reaction of oxidation-reduction, electrons flow from zinc to copper through the external circuit, producing a potential difference (voltage).
- 🔌 Electrodes: metal parts (Zn and Cu) where chemical reactions occur.
- 💧 Electrolyte: ionic conductive medium (citric acid or potato juices).
- ⚡ Voltage: “electrical ”thrust"; several cells in series add voltages.
🔬 Scientific method: your plan of attack.
- Observation: Some fruits and tubers conduct electricity.
- Question: Which combination (lemon vs. potato, electrode size, number of cells) produces more voltage?
- Hypothesis: If I use fresh lemons and large Zn and Cu electrodes, then I will get higher voltage than with potatoes.
- Experimental design: Keep temperature and connection type constant; change only one variable at a time.
- Experimentation: Build cells and measure their voltage with a multimeter. Record data in tables.
- Analysis: Compares averages and graphs; evaluates variability.
- Conclusion: Accept or refute the hypothesis and justify with your data.
- Communication: Present your poster with outline, photos and results.
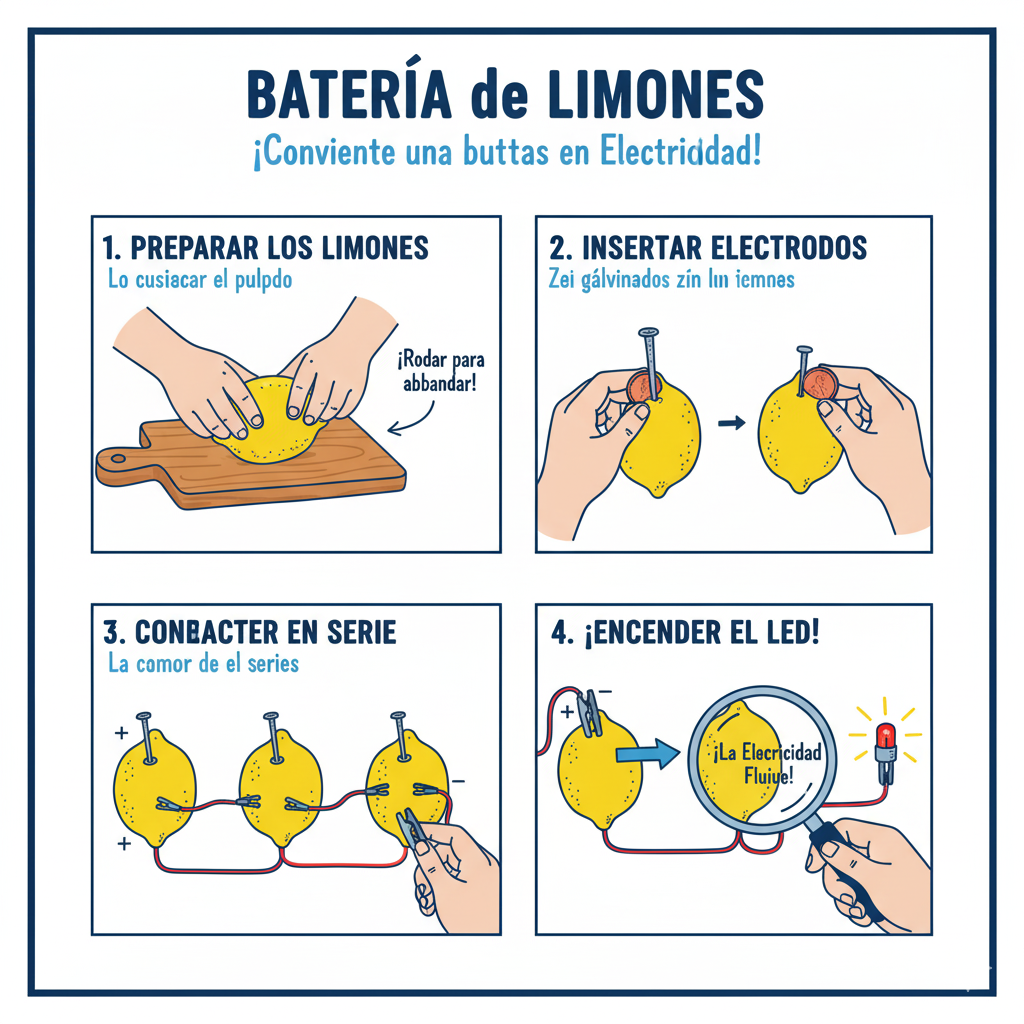
🧩 Graphic description of the model or assembly.
This is what a cell with a lemon (or potato) looks like. Connect several in series to increase the total voltage.
(+) wire (-)
Cu plate ─────┐ ┌─── Zn nail.
│ │
.-''''-. │ │
.' 🍋 '.│ │ │ LEMON (acid electrolyte).
/ Cu Zn \│ │ │
| || |
\ / │ │
'._ _.' │ │
'---' └──┘
(towards LED or multimeter)
🛠️ BOM with smart options
| Material | Economic | Standard | Professional |
|---|---|---|---|
| Fruit/acid tuber | 1-4 medium lemons or potatoes | 6-8 lemons/potatoes of the same size | Dozen calibrated lemons/potatoes |
| Copper (Cu) electrode | Copper coin/ribbon | Copper plate/laminate 1-2 cm² Copper plate/laminate 1-2 cm² Copper plate/laminate | Laboratory grade copper electrodes |
| Zinc (Zn) electrode | Galvanized nails (zinc plated) | Commercial zinc strips | Pure zinc electrodes |
| Cables | Cheap alligator clip cables | Set with quality insulation | Shielded banana-crocodile set |
| Measuring / Loading | Red LED and 220-330 Ω resistor | Basic digital multimeter | True RMS multimeter / data-logger |
| Support | Cardboard or plasticine | Small Protoboard | Mounting plate with fasteners |
| Others | Insulating tape, marker | Tape + cutter + gloves | EPP kit (goggles, gloves, mat) |
🧭 Step-by-step guide: your adventure map
1) Prepare the cells
Gently roll each lemon/potato on the table to soften it without breaking the skin. Make two small incisions to insert copper y zinc, spaced about 3-4 cm apart.
2) Insert the electrodes
Insert the electrode Zn (galvanized nail) and the Cu (coin or plate). They should not touch inside the fruit/tuber.
3) Connects in series
Join the Cu of the first cell with the Zn of the second, and so on. Leave free a Cu at one end and a Zn on the other to measure the total voltage.
4) Measure voltage
With a multimeter in V DC, the red tip on the Cu (positive) and the quarter note in the Zn free (negative). Record the value in your data sheet.
5) Turns on an LED
Connects a LED in series with a resistor (220-330 Ω). The long side of the LED goes to the positive pole (Cu). If it does not light, increase the number of cells.
6) Control variables and repeat
Try with lemon vs. potato, Change electrode size, change electrode size, or add more cells. Record each change and measure 3 times to average.
🎪 Prepare your presentation for the fair.
- Clear poster: Eye-catching title, photo of the assembly, ASCII schematic and actual diagram.
- Interactivity: It allows the jury to measure live voltage or compare lemon vs. potato.
- Phrases that impress: “Our battery is a galvanic cell: zinc is oxidized, copper is reduced.”.
- Key data: Voltage per cell, number of cells, resistance used, conditions.
📎 Annex A - Data recording template.
| Test | Fruit/Tubercle | # Cells | Electrode size | Voltage (V) | Comments |
|---|---|---|---|---|---|
| 1 | Lemon | 4 | Medium | ||
| 2 | Potato | 4 | Medium | ||
| 3 | Lemon | 6 | Grande |
✅ Annex B - Check list
- Cleaned metals (Cu and Zn)
- Correct series connections
- LED with series resistor
- Dated and timestamped data
- Safety standards applied
- Plan to repeat and verify
🔎 Recommended sources
Introductory resources on galvanic cells and electrochemistry (to be expanded):
- General chemistry textbook (electrochemistry chapter).
- Educational encyclopedias and learning platforms with articles on batteries and batteries.
- School laboratory manuals with lemon battery practices.