
Create a Chemical Rainbow!
Your own pH Indicator with Red Cabbage. Discover the acidic and basic secret of the things around you. Science has never been so colorful!
🎯 Your Scientific Missions
- General Objective: You will build a homemade 100% pH indicator using red cabbage and use it to classify common household substances as acidic, basic or neutral.
- Personal Objective: Challenge yourself to think like a real scientist! You'll experiment with confidence, record your findings, and wow everyone at the science fair with a visually stunning and easy-to-understand project.
🌍 What is pH? The Secret Superpower of Substances!
Imagine that all liquid substances have a personality: some are «acidic» like lemon juice 🍋 and some are «basic» (or alkaline) like soap 🧼. The pH is simply a scale that measures how acidic or basic a substance is - it goes from 0 (very acidic) to 14 (very basic)!
- Acids (pH 0-6): They usually have a sour taste. Think vinegar or orange juice.
- Neutral (pH 7): The perfect middle ground, like pure water 💧.
- Bases (pH 8-14): They are usually slippery to the touch. Baking soda or cleaning products are good examples.
Our red cabbage is a wizard of chemistry. It contains a molecule called anthocyanin, It changes color according to the pH of the liquid it is in, making it a perfect natural indicator!
💡 Did you know that...?
Anthocyanins are not only in red cabbage - they are responsible for the red, purple and blue colors of many fruits and flowers such as blackberries, cherries and petunias! Nature is full of pH indicators.
🔬 Your Plan of Attack: The Scientific Method.
Every great scientist follows a plan. This will be yours:
- Observation: You have seen that some substances, such as lemon, are sour, and others, such as soap, are bitter or slippery. They look different.
- Question: Could you use something natural, such as a vegetable, to visualize these differences in a colorful way?
- Hypothesis: «If I mix red cabbage juice with different homemade substances, it will change color. Reddish/pinkish colors will indicate that the substance is acidic, and blue/green/yellow colors will indicate that it is basic.».
- Experimental Design: This is the fun part! You will follow the steps in this guide to prepare your indicator and test your substances.
- Analysis of Results: You will note all color changes you observe in your data table.
- Conclusion: You will compare your results with your hypothesis. Did it come true? What did you learn? You are now an expert!
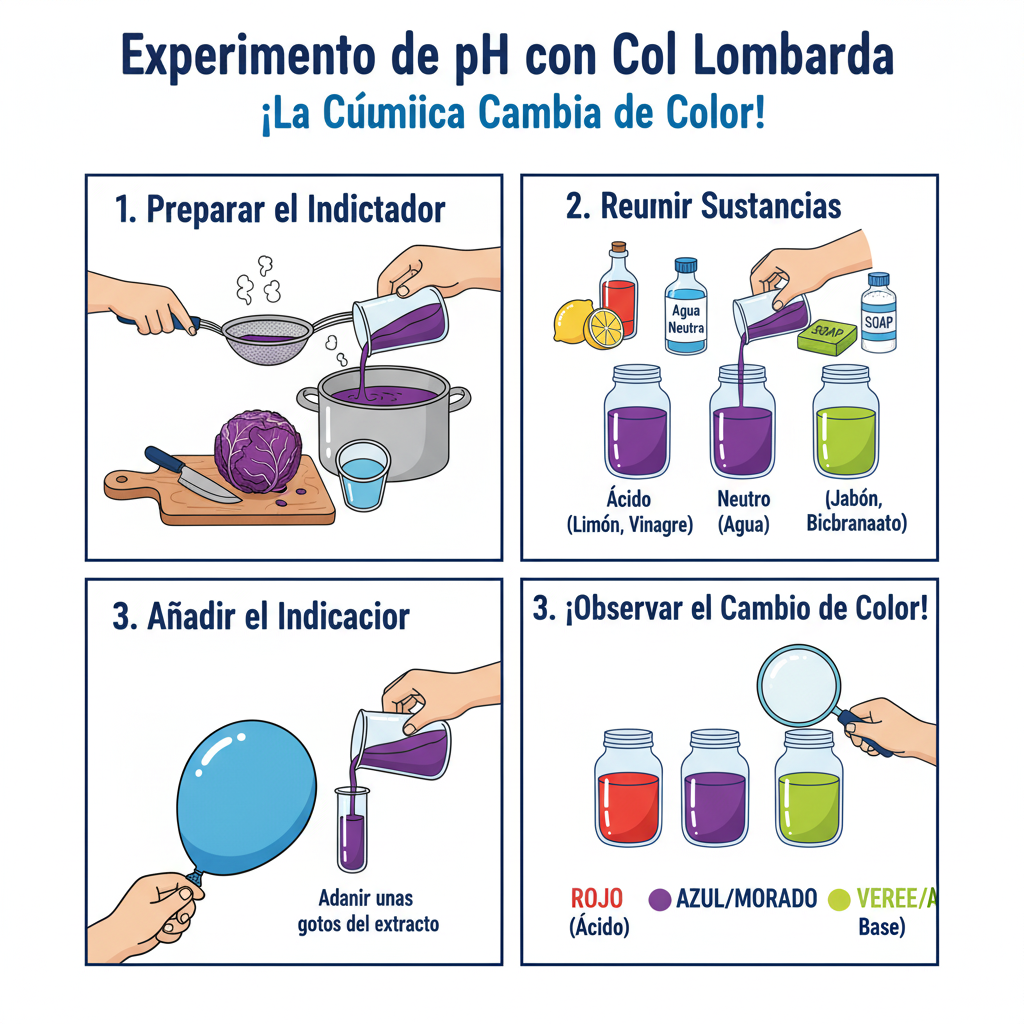
🧩 This is what your Laboratory will look like
Your workstation will be a colorful row of containers. You will have one large container with your purple «magic potion» (the cabbage extract) and several smaller beakers or test tubes, each with a test substance. By adding a few drops of your indicator, each beaker will reveal its secret in spectacular color.
Your Indicator Your Samples
(Purple)
____ _ _ _ _
| | -----> | | | | | | | |
|____| |_| |_| |_| |_|
(Red cabbage) (Lemon)(Water)(Soap)(Bicarb)
⚠️ Watch Out Here! Caution Zone
Although this is a home experiment, safety first. Ask an adult to help you cut the cabbage and, above all, to handle the hot water. Wear safety goggles if you have them and never, ever taste the substances in the experiment.
🛠️ Scientific Arsenal: Materials List
You don't need a NASA lab - here are options for every budget!
| Material | Economic Option | Standard Option | Professional Option |
|---|---|---|---|
| Indicator | Half red cabbage | One whole red cabbage | Anthocyanin extract powder |
| Test vessels | Glass or clear plastic cups | Plastic test tubes with rack | 50ml beakers |
| Filter | A kitchen strainer | Gauze or coffee filter cloth | Laboratory filter paper |
| Heat source | Pot with hot tap water | Pot on the stove (with supervision) | Laboratory heating plate |
| Test substances | Lemon, vinegar, baking soda, water | Add milk, soda, liquid soap | Add antacid, ammonia (with caution). |
🧭 Your Adventure Map: Step by Step
- Prepare the Magic Potion (20 min): Ask an adult to cut half a head of red cabbage into small pieces. Place them in a large, heat-resistant bowl. Bring water to a boil and carefully pour it over the cabbage until it is covered. Let it sit for at least 15 minutes - you'll see the water turn a deep purple color!
-
Filter your Indicator (5 min): Use a strainer to separate the cabbage pieces from the liquid. That purple liquid is your pH indicator! Let it cool.
⭐ Pro Tip
For a more concentrated and darker indicator, you can blend the cabbage with a little water instead of using hot water - just be sure to filter it very well!
- Organize your Lab (10 min): Place your test containers in a row. In each, put a small amount of the substances you are going to test (e.g., a teaspoon of baking soda dissolved in a little water, a splash of lemon juice, etc.). Label each container so you don't get confused.
-
The Moment of Truth (15 min): With an eyedropper or teaspoon, add your cabbage indicator to each of the test substances a little at a time. Watch the magic and note the colors that appear in your data table!
🚨 Scientist Alert!
If you decide to use cleaners such as ammonia or bleach, do so in a well-ventilated area and wear gloves. Never mix bleach with ammonia, as it produces very dangerous fumes!
🎪 Let's Shine at the Science Fair!
Your experiment is visually great. Now, let's make your presentation cool too!
Your Poster
Divide it into clear sections: Catchy Title, Question, Hypothesis, Materials, Procedure (with pictures of you doing the experiment), Results (the most colorful part!) and Conclusion.
Interactive Ideas
Don't just talk, demonstrate! Have your indicator and some safe substances (lemon, baking soda, water) handy. Invite the judges or visitors to add a few drops and watch the color change live - they'll love it!
Phrases to Impress
- «This experiment demonstrates the principle of acid-base indicators using anthocyanins, a natural pigment...»
- «As you can see, the results validate my hypothesis: acidic substances veer to red tones, while basic substances to green and blue tones.»
- «This homemade method is a sustainable and accessible way to introduce fundamental chemistry concepts.»
📎 Attachments: Extra Tools!
Data Recording Template
| Substance Tested | Predicted Color (Hypothesis) | Observed Color (Result) | Classification (Acid/Basic/Neutral) |
|---|---|---|---|
| Lemon Juice | Pink/Red | ||
| Water | Purple/Blue | ||
| Baking soda with water | Green/Blue | ||
| Vinegar | Pink/Red |
Final Checklist
Recommended Sources
Want to know more: curiosity is the engine of science! Explore these sites:
- Khan Academy (Chemistry)
- National Geographic Kids
- Popular science YouTube channels such as «Plato's Robot» or «CdeCiencia».

